
INVESTOR OPPORTUNITY
The medical sterilization market represents a billion-dollar industry disruption opportunity.
The VHP sterilization equipment market is dominated by a few lethargic companies offering archaic “dumb” technology.
It’s time to think beyond conventional VHP sterilization.
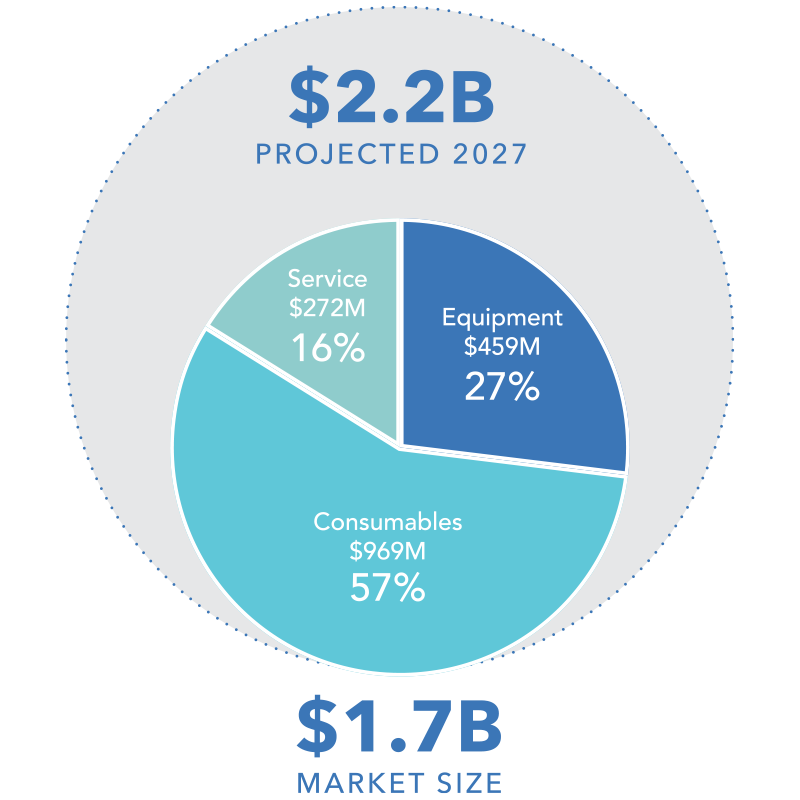
Sterilucent will disrupt the stagnant $1.7B medical VHP sterilization market by creating an entirely new category that will redefine standards of performance, user experience and patient care.

FACT:
Conventional VHP sterilization is old technology.
The failure of conventional VHP technology to keep up with the challenges of modern healthcare is costing healthcare providers hundreds of millions of dollars and putting patient lives at risk.
Sterilucent’s patented technology innovations represent a $1.7 billion market disruption opportunity.
Problems with conventional VHP sterilization
Time-wasting failed cycles
The #1 cause of aborted cycles is inadequate moisture detection and drying capability.
Inefficient use of sterilants
Injecting fixed amounts of sterilant is expensive, inefficient and environmentally unsound.
High operating costs
Costs include failed cycles, expensive consumables, surgical delays, high service costs
Damages expensive instruments
Millions are spent by hospitals on repair or replacement of new or advanced instruments.
No analytics, no control
Conventional tech can’t actively measure and adjust throughout the cycle.

Can't sterilize many instruments
Conventional technology can’t be used on many advanced surgical devices and materials.
OUR SOLUTION

Introducing the dawn of a bold new era:
The world’s first smart, analytics-driven sterilizer
Sterilucent will energize the stagnant $1.7B medical VHP sterilization market by creating a disruptive new category that will exploit conventional VHP’s out-dated technology.
Driven by cutting-edge innovations in proprietary sensing and data analytics technology, Sterilucent will deliver game-changing performance never before thought possible.
A LARGE AND GROWING MARKET
What’s driving 7% annual growth?
DEMOGRAPHICS
- Expanding patient and geriatric population
- Rapid increase in chronic diseases
- Increase in unhealthy lifestyles
- Rising life expectancy
RISING INFECTION RATES
- Growth in anti-microbial resistance
- Rapid increase in hospital-acquired infections
- Hospitals moving away from disinfectants
INCREASE IN SURGERIES
- Increasing number of hospital surgeries
- Increasing use of advanced surgical devices
- Growth in outpatient surgery and endoscopy
- Growth in clinics performing surgery
REGULATORY
- Increasingly stringent regulatory policies
- Govt. recommends use of better, safer equipment
- Govt. pushing reduction of infection rates
- Favorable reimbursement for outpatient surgery
ENVIRONMENT
- EtO is being permanently phased out
- EPA and OSHA are driving shift from EtO
- EtO is carcinogenic
- Concerns with sterilant safety and usage precautions
ECONOMIC PRESSURES
- Rising cost of sterilizer consumables
- Rising cost of expensive instruments
- Pressures to increase staff productivity
- Need to replace high number of aging sterilizers
- Need for less expensive service contracts
HOW INVESTORS WIN

Clear pathway to success
With Sterilucent’s strong fortress of patent protection and agile technology platform, the company is strategically well-positioned to aggressively compete in a stagnant industry that’s ready for disruption.
1
FDA Approval
Projected Q2, 2024. Results in immediate 2x increase in company valuation
2
Opportunity for early exit
Based on strategic fit with suitors awaiting our FDA approval
3
begin commercial sales
Q2, 2024, establish a differentiated beachhead in the $1.7B marketplace
4
build on beachead success
Opportunity to grow company to
8x valuation
Attractive Investment Opportunity
• Product development is 95% complete
• Majority of investor funds used for commercial prep
• Regulatory path is straightforward
• Operational and technical risks have been resolved
• Highly capable leadership
• Customer-validated
Special Investment Offer
To provide extra incentive, Sterilucent is offering a preferred investment vehicle. Learn more by requesting additional investor info below.
Sterilucent is striving to attain attractive investor returns based on achieving critical milestones. Opportunities and risks are further defined in additional investment information and disclosures, available upon request.

About Sterilucent
Sterilucent was founded in 2004 by David C. Bell, a visionary business development leader with over 40 years of worldwide experience in the development of new technologies and bringing them successfully to market. Sterilucent’s initial funding was secured from the National Institute of Health (NIH) with additional interest from the Department of Defense (DoD). Continued support from NIH and angel investors provided additional funding to allow Sterilucent to complete development of its groundbreaking technology.
The company’s mission is to create long-term corporate growth and sustained stakeholder value by disrupting the multi-billion dollar VHP medical sterilization equipment market.
